The title of this presentation is an acrostic that lists the six elements needed for life: Sulfur, Phosphorus, Oxygen, Nitrogen, Carbon and Hydrogen.

If you read it backwards, it also says “SPONCH.” Image via Wikipedia: “The relative atomic diameters of carbon, hydrogen, oxygen, nitrogen, phosphorus, and sulfur.”
Life on this planet has a very old history. It has emerged from a primeval soup; cooked with the Sun for the last 4.6 billion years.
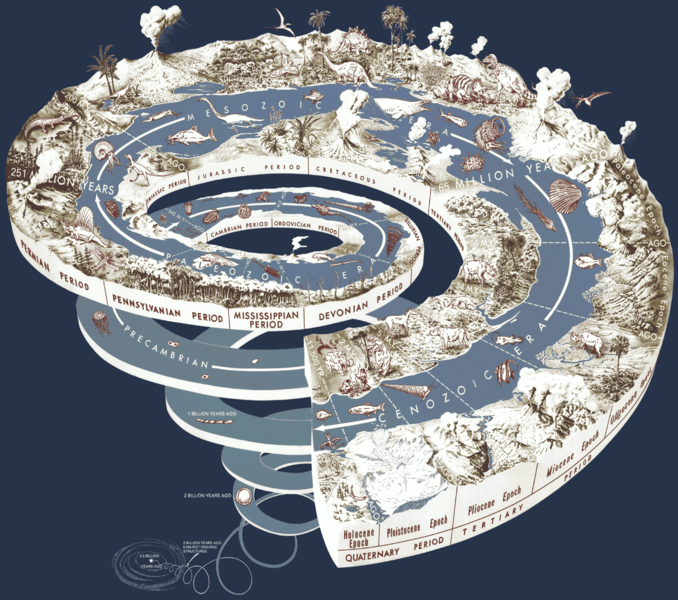
Where else do we see the spiral? A brief essay (150 words), about the pattern of the spiral in nature, can be accepted as a participation on the blog (10 points).
“Earth’s prebiotic oceans—different from their modern counterparts—would have formed a “hot dilute soup” in which organic compounds could have formed. J.D.Bernal, a pioneer in x-ray crystallography, called this idea biopoiesis or biopoesis, the process of living matter evolving from self-replicating but non living molecules,”
Ancient oceans were very hot. And barren. No life. But they were huge cauldrons; with the proper conditions to cook with CHNOPS. 4.6 billions of years ago, amino-acids–building blocks of life–were emerging. But how these elements happen in the first place? The subject is a matter of heated debate in the modern scientific community, but recently NASA came across this interesting piece of data:
In March, 2009, researchers at NASA’s Goddard Space Flight Center in Greenbelt, Md., reported the discovery of an excess of the left-handed form of the amino acid isovaline in samples of meteorites that came from carbon-rich asteroids. This suggests that perhaps left-handed life got its start in space, where conditions in asteroids favored the creation of left-handed amino acids. Meteorite impacts could have supplied this material, enriched in left-handed molecules, to Earth. The bias toward left-handedness would have been perpetuated as this material was incorporated into emerging life.
Another elegant approach towards the origin of life, in an experiment 60 years old this year:
Asteroids could’ve brought amino acids. Hot, primordial, oceans boiled (remember the emergent properties of water); lightning storms cooked electric. Are we made out of star stuff? Where these elements the result of The Big Bang?
[Put on your headphones and Google—or better yet: retrieve from Youtube–the song Solstice (Current Value Remix for Seniors; original version for 10th grade). Listen to the song. Look for the lyrics. Write a short essay (100 words) on the song (10 point value; it can replace a blog participation).]
On this spinning rock, macromolecules have emerged. And they are strange: they can replicate. Information is carried in their chemistry—in their molecular arrangements—in their bonds—5-Carbon sugars.
DNA replicates. It can be read, reread, edited, transcripted, mutated, sliced. All this happens because amino-acids—23 of them—combine in ways that make horns, hemoglobin, muscle tissue, etc.
Plants can do this in their cells; the same mechanics are embedded in our own DNA. Thanks to these remarkable properties–possible only through the chemical interactions between CHNNPS elements–plants pumped a very important element to the atmosphere; these elements ignite flames; they were believed to be phlogiston (the prefix “phlo” refers to flammable).
Oxygen is highly reactive as well, and it’s responsible for trillions of controlled explosions going on in our cells; right now.
Oxygen–versatile element–makes fire possible, it makes Cellular Respiration possible.
Another element famous for its versatility is Carbon. It can combine in its pure form in various ways, forming what we know as allotropes. These can be bucky balls–they travel through space–; graphite (your pencils are made of this); graphene; diamonds; even whole planets.

An illustration of 55 Cancri e shows a surface of mostly graphite surrounding a thick layer of diamond. Illustration courtesy Haven Giguere, Yale
A lot more can be said about the origin and nature of life. But this presentation aims to spark your curiosity about biochemistry, abiogenesis, and, to some extent, cosmology–we are part of the Cosmos.
This video is by science commentator and physicist, Neil Degrasse Tyson; a good coda–ending–to this blog post, which will be revisited– many times–during the school year:
_______________________________________________________________________________________________________
Important Information:
This blog post is for both: 10H and AP Biology. The content of this presentation will be evaluated on future assessments—i.e. tests, quizzes, quizams, etc. Textbooks for this presentation are: Modern Biology, Campbell 7th through 9th, and Study Guide for Modern Biology.
1oH:
Unit I: The Foundations of Biology
Chapters 2 (Chemistry of Life) and 3 (Biochemistry)
The film presented on class, Nova: Hunting for Elements, can be used to review concepts discussed here.
Homework: Chapter 3 of your Study Guides for Tuesday 10th of September.
AP Bio:
Unit: Intro to Biology.
Chapters 3 (The Chemical Context of Life) and 4(Carbon and The Molecular Diversity of Life).
Labs I (Bonds and models) and II (Emergent Properties of Water) are important for the discussion of this presentation. Concepts 3.1, 3.2, and 4.1 are aligned with this blog post.
Homework: One participation—comments, replies, etc.—on this blog post. (Refer to the rubric given by the teacher on how to comment).


Carbon is such a versatile element because it has 8 electrons that allow it to stick to many other elements or create compounds. This element is able to create a chain which permots other elements to branch out. Carbon is also known as the backbone of life since it’s one of the building blocks of life. Thanks to organic chemistry, which is science that studies all that has to do with carbon, we know the importance and versatility of carbon. (Adriana Berrios Surillo)
LikeLike
Carbon is also one of the most abundant elements in the universe. And all of the carbon that is present in our bodies, animals, rocks, fossil fuels, etc., was once—billions of years ago—forged in the furnaces of ancient and distant stars. Thus, we are made out of «star stuff». Good comment, Adriana.
LikeLike
When we first studied C.H.N.O.P.S., I didn’t quite understand why phosphorus and sulfur were such vital elements for earthly life. But, thanks to our study of organic compounds, I now understand that a variety of organic compounds have phosphorus (such as the ones we studied in class): phospholipids, ATP, many amino acids… Even though phosphorus was clarified to me, I still didn’t know why sulfur was important, so, as any biologist would do, I researched! I found out that sulfur is consumed by humans in the form of amino acids and proteins, as shown on this link: http://www.lenntech.com/periodic/elements/s.htm
Even though the sulfur we are familiar with isn’t toxic, simpler versions of it can be harmful to humans and animals alike. If you are interested in this topic, I suggest viewing the information provided by the link I posted. 🙂
LikeLike
Mankind has known about sulfur for millennia. So that means that, even without the modern tools of scientific research, we’ve been on to something for a very long time. Great research, Arnaldo.
LikeLike
Before watching this video I was unaware of how useful graphine was. I never would have thought that graphine played such an important role in our daily lives. Thank you for this wonderful video, it was very ineteresitng and I learned a lot from it.
LikeLike
Just imagine what will happen in a not too distant future. This carbon structure holds a lot of promise, especially in terms of our energetic needs.
LikeLike
Hydrogen can bond with any electronegative atom of another element, these bonds are called covalent bonds because they share electrons. These types of bonds can also create a type of hydrogen bond, which are weak between compounds that contain hydrogen.
LikeLike
Hydrogen bonds are very important for the sustainability of life on Earth–especially if we consider how water can easily change its states of matter.
LikeLike
Pingback: Cell biology and art. | The Hypertextual Lounge
I will admit that the first few times reading this text i didn’t fully comprehend it, so i searched for the best possible way to help me broaden my understanding of this topic, & i found one. Following the constant topic of the universe’s infinitely huge & infinitely minuscule existence, i found that the knowledge of these ingredients of life is essential to every art of science, and, really, life. The link i found & this blog post also helped me imagine how it’s all linked together: these may be the eternal elements of life, but there are many other things that have to contribute and work together to makes this whole “Life” thing work; the necessary temperature, the existence of liquid water, the correct distance of the Sun from the planet. It all goes together.
http://m.livescience.com/32983-what-are-ingredients-life.html
LikeLike
The Kepler telescope has identified rocky planets orbiting the circumstellar habitable zone—CHZ, or ‘goldilocks zone’—of a red dwarf hundreds of times less bright than our star. There is a chance that this planet is one of the 500 million possible ‘twin earths’ that astronomers look for. There is a chance that this planet has pools of (liquid) water, the universal solvent.
LikeLike
And how far away would one of those ‘Earth twin’ planets be?
For the existence of life, liquid water is essential to go with the other necessary elements & materials so this is a great venture/discovery in astronomical science.
LikeLike
This particular exoplanet is more than 20,000 light years away.
LikeLike
That’s truly incredible, especially when you consider the possibility of life existing in these planets. As I read your comment, I was confused by the term ‘goldilocks zone’ since it was a name I’d never heard of, and I couldn’t understand why this was considered the habitable zone. I googled it, and I discovered that the Goldilocks region is an area of space that is considered habitable because the planets in it are just the right distance from their home star so that their surface is neither too hot nor too cold. In between, the conditions are just right in order for liquid water to remain on the surface of the planet without freezing or evaporating out into space. For more information, check out the following link: http://science.howstuffworks.com/other-earth1.htm
I also found out how this region received its name. It comes from the children’s story, “Goldilocks and the Three Bears.”
“This porridge is too hot,” Goldilocks exclaimed.
So she tasted the porridge from the second bowl.
“This porridge is too cold.”
So she tasted the last bowl of porridge.
“Ahhh, this porridge is just right!” she said happily.
And she ate it all up.
Found in http://science1.nasa.gov/science-news/science-at-nasa/2003/02oct_goldilocks/
LikeLike
Life is a tricky business. Many things have to happen: liquid water, reactive elements that yield energy and serve as structure—a lot of requirements. So it is fair to say that life is not a common event in the Universe.
Google the «Drake Equation» and check out how probable life can be. This equation even predicts the possibility of not just life in other star systems, but ‘intelligent’ life, civilizations that could, if given the proper circumstances, communicate with us.
Great links and great comments, Isabella.
LikeLike
Carbon is so versatile because it can easily combine with the atoms found in various other elements. These other elements are resistant to such changes. The atoms in carbon can form chains in the structure of single, double and triple bonds. As studied in class there are types of bond such as covalent, ionic , and hydrogen bond that bond atoms together. Hydrogen bond are very important to life because hydrogen bonds allow water to remain liquid at room temperature when it would otherwise be a gas. It also allows a number of nitrogen and oxygen containing compounds to pass easily through the body.
LikeLike
To think that all of our carbon atoms in our bodies were cooked “in the furnaces of ancient stars.”
LikeLike
I found this blog post very interesting due to the fact that all life depends on six vital elements. The one that impressed me was carbon. As we all know carbon is the “backbone of life”. I did further research on carbon that stated that carbon is the backbone of life due to the versatility of the element and its ability of merge with other carbon atoms forming long chains of carbon as a base for other atoms to bond with this element. That is carbon, now for nitrogen, oxygen, and hydrogen. These elements are good because they are abundant everywhere (within reason). They also exhibit acid-base effects, which allows them to bond with carbon to make amino acids, fats, lipids and the nucleo-bases from which DNA and RNA are built. Sulfur provides electron shuffle (generate energy for survival by transporting electrons to nearby mineral surfaces) and basically, with their surplus of electrons, sulfides and sulfates help catalyze reactions. Finally, phosphorus is vital to metabolism, because polyphosphate molecules such as ATP (adenosine triphosphate) are able to store a huge amount of energy in their chemical bonds.
For more information access this link:
http://www.livescience.com/32983-what-are-ingredients-life.html
LikeLike
Just imagine the possibilities of life out there, in space; given that carbon is the 6th most common element in the universe.
LikeLike
In life, CHNOPS is very important. Without these elements (carbon, hydrogen, nitrogen, oxygen, phosphorus, and sulfur), life wouldn’t be possible. We saw in the movie and disscused in class, how each element is used and how important they are. For example carbon has a special characteristic which allows it to bind to other atoms, making it a great building block for organic molecules. On the other hand, sulfur is a great catalyst. Thirdly, phosphorous is an important molecule in order for metabolism to take place. Hydrogen and oxygen together make water which is vital for us.
As you can see it is very necessary that we know what CHNOPS is and how important it is to us.
http://m.livescience.com/32983-what-are-ingredients-life.html
LikeLike
I encourage you to Google more information about carbon, especially graphene—a carbon «allotrope» that promises big changes in information technology, bioengineering, green technology, etc.
LikeLike
It is amazing how DNA can do so much being a non-living molecule. It can copy important information and can duplicate itself, without it life would not be possible, traits would not exist. it is unbelievable how a molecule without a brain, nor any part that gives command, can do so many important processes in a incredibly complex body. DNA in viruses can be putted in a sealed jar for years and when you take it out it can still cause the disease. Being inert (not moving, not breathing, no respiration and no reproduction) the DNA can keep its characteristics and still do its job when it return to the environment. It is really a fascinating molecule which controls a big part of life. A single strand of DNA can not be alive by its own. A cell can not be alive without the DNA and the organism can not be alive without the cells that depend on this microscopic molecule call Deoxyribonucleic acid. In conclusion DNA is a really powerful molecule which has a huge effect on life.
LikeLike
You made a very good analogy, Josué—the virus being a ‘jar’ that stores deoxyribonucleic acid.
LikeLike
Before visiting this blog and studying the chapter I did not know how important carbon was to life. I had previously thought that oxygen was the most important element for life. I now know that oxygen is the most abundant but carbon is the “backbone of life”. I also learned that elements such as phosphorus and sulfur are very important for life. I found interesting how graphine could purify water and be so useful. In this picture it shows how many bonds or how reactive is each element is and gives some examples of where it is found. https://www.google.com.pr/url?sa=i&rct=j&q=&esrc=s&source=images&cd=&cad=rja&docid=ZrEKXLOQMpDs7M&tbnid=joHAtN_upz835M:&ved=0CAUQjRw&url=http%3A%2F%2Fwww.biologylessons.sdsu.edu%2Fta%2Fclasses%2Flab2%2FmapTG.html&ei=wkdHUvPBFIqA9QTv74HYBA&bvm=bv.53217764,d.eWU&psig=AFQjCNGSrouS1kDngwPr–qjdev2xoQ4ZQ&ust=1380489406726750
LikeLike
Buy the time your grandchildren have your current age, I expect that graphene will be used I almost every important aspect of human experience. The next few decades will be exiting times in science. Let’s hope scientists have proper ethical considerations.
LikeLike
I accidently put the link for the website not the photo. The photo is in the website if you scroll down a little bit:/ Sorry
LikeLike
No problem, Carla. Thanks for the correction.
LikeLike
I would like to comment about the last video by Neil Degrasse Tyson. I thought this video was amazing. I, as he mentions in the video of other people, felt we as humans were very small in the univers but he has showed me another way of thinking. It is true that were are all connected since we all came from the same stars. These exploding stars, providing all things necessary for life especially the SPONCH elements, since without them we couldn’t be able to survive. The SPONCH elements also show how all living things are in another way alike because no matter what we all need these elements. These two things show how we are all connected. I also found this viedo that presents how life is just a cycle and that we are all the same in a weird sort of way:
LikeLike
I’m sorry, I tried to put the link put the video showed up directly…
LikeLike
I encourage you to Google Carl Sagan, a scientist, writer, and humanist that Neil DeGrasse Tyson respects a lot. Actiually, DeGrasse Tyson continues his legacy as a science communicator; putting lofty scientific concepts in plain English. Sagan was also the creator of a wildly popular science series during the 1970’s called Cosmos—based on one his most popular book. DeGrasse Tyson will be the new host of a remake of this series, updated for the 21st Century.
LikeLike
thanks:) I found really interesting that since he was a little kid Carl Sagan had interest in the universe, wondered and questioned a lot of things, something that would help him for the rest of his life. I also found interesting that he remembers going to the New York World’s Fair as one of the greatest experiences as a child and that although his parents bought him sets and books of diferrent things he always had a deep interest in space.
LikeLike
I’m sure that many of those books were science fiction.
LikeLike
In life there are 92 elements that are known, 25 of this that are essential for life and six of these elements termed to be the “building blocks of life.” They are, in order of least to most common, sulfur, phosphorous, oxygen, nitrogen, carbon, and hydrogen. The remaining 19 elements are defined as trace elements, which are needed in small quantities, and are usually required by specific proteins to function.
LikeLike
Are there any other candidates for life other than carbon? I suggest you do research on what scientists have discovered on Saturn’s biggest moon, Titan. Polypropylene, a common polymer found in man made plastics, is abundant in this moon’s atmosphere. Could there be life on Titan?
LikeLike
Before researching and reading these blogs I didn’t have so much knowledge about this evolution and changes in life. The image of the spiral shows that life has evolved during time and in an exagerated way. Life in the past or in the beggining was nothing like life as we know it. Most of the evidence for an ancient Earth is found in the rocks that form the crust. This historical spiral records important scientific and historic events that have occured through time. This layers have buried within them animals and plants that evolved from organic structures that came to exist 3 billion years ago.
The link I posted is a short movie of life’s evolution
http://m.youtube.com/watch?v=MKn0xWM7qh0&desktop_uri=%2Fwatch%3Fv%3DMKn0xWM7qh0
LikeLike
Great video.
The spiral is ubiquitous—appearing, or found everywhere—in nature; we see them in the shape of our galaxy, in the conch of snails, and ancients have used the spiral as a symbol for rebirth, evolution, and even infinity.
In the case of this graphic, the spiral is used to illustrate the property of ’emergence’. Google this word and try to establish a relationship between ’emergence’ & ‘evolution’.
LikeLike
I didn’t know or imagined the importance of CHNOPS until this year when we discussed it in class. Who knew that Carbon, Hydrogen, Nitrogen, Oxygen, Phosphorus, and Sulfur were the 6 most abundant elements of life and known to human kind. Life as we know it takes dramatically different forms, but all organisms are but from this six essential elemental ingridients. In this elements there’s carbon which is know as “the backbone of life.” That detail makes it obvious that is very important in life.
In realclearscience.com or livescience.com it tells you why are these 6 elements so important in our everyday life.
LikeLike
Both websites are very good. It is convenient for life to use these six elements; they are very common in the universe, and very reactive.
LikeLike
I looked for information on Carl Sagan and Neil DeGrasse and tought it was such a cool idea as Neil says ” We have a duty to try and inspire those we teach just as we were inspired by those who taught us”. Neil was really inspired by Carl and as he says in he link I am posting he had a great influence by him so, he wants to be the same to others that want to know about the universe like he once did. He will be the host in a remaking of Carl’s series Cosmos to commemorate him.
LikeLike
For me Carl Sagan is a constant source of inspiration. He died of cancer in 1997, but his legacy continues to inspire scientists, educators, and students—unfortunately, politicians are not included in this list.
LikeLike
Before reading this blog I didn’t know the importance of these six elements except for carbon. In class while discussing the chapter, we talked about the importance of carbon. Carbon is the most important element and is the basis of life. It is known as “the backbone of life.” I wanted to learn more about the other five elements so I searched for more information. I found that out of the 92 natural elements, 25 are essential for life and six of these elements are considered the building blocks of life. These six elements are known as CHNOPS (carbon, hydrogen, nitrogen, oxygen, phosphorous, and sulfur).
LikeLike
Even when life a complex and rare event in the universe, it is quite an amazing fact that it is possible with just six elements.
LikeLike
After watching the graphene video I was very surprised to see the different advantages this material could offer to our society. As the video says, its conductivity could help with solar powering. This has become a new trend in technological advances now a day due to the dependency people have created with electricity. Electricity’s cost if going up everyday and it is playing a huge part in the economic crisis of today. If scientist use graphene to create new and more efficient devices that would help people change the way in which they receive the electricity needed for their home appliances, maybe people would have more money to pay debts and the economic crisis will stop increasing.
Graphene could also play a huge role in countries that have problems accessing clean water supplies. Africa is one of the most recognized countries with this problem. Many organizations and foundations have been created with the goal to help this people have clean water in the cities. Maybe this same people could invest in making graphene the main resource used to make products that will help ease this problem.
It is very interesting how biology and chemistry could function together and help solve problems in society. As a scientist, I think it is great to make that connection between society and the research he or she does to help other people in the way they live.
Here is a recent article I found about an interview CNN did with Andre Geim about graphene:
http://edition.cnn.com/2013/10/02/tech/innovation/graphene-quest-for-first-ever-2d-material/index.html
LikeLike
I’m fascinated by graphene and all of its possibilities. A lot of high hopes for this single atom thick, bi-dimensional structure.
LikeLike
I was unaware of how useful graphine was and how it could help us in our daily lives. I hope that scientist start making things with graphine soon.
LikeLike
Scientists, corporations, and countries like China, England, Singapore, etc., are betting a lot on the possible applications of this technology–especially in the next 5 to 10 years.
LikeLike
Learning about CHNOPS really helped me understand this material. I had no idea that that those six elements were so important for life to be possible. What shocked me about this blog post was the fact that scientists found left handed amino acids on a meteorite and this suggests that left handed life got its start in space and then arrived on earth. I read this article and it helped me understand more about this theory. http://www.space.com/20888-earth-life-amino-acids-asteroids.html
LikeLike
What shocked you shocked a lot of scientists. This discovery put a new perspective to the what can happen with CHNOPS in outer space–especially with C,N, and, O, essential for macromolecules such as proteins, DNA, carbohydrates, lipids, etc.
LikeLike
This post on “SPONCH” or “CHONPS” i believe catched the attention of my peers and myself very highly. I was unware of the importance of all of these elements on our daily life. Just like graphine, these six elements play an important role on Earth. Although there are 92 elements, 25 are important for life and out of the 25 these 6 are essential. I found this link which explains thoroughly what we discussed in class about “CHNOPS”. The basis of life is Carbon, “the backbone of life”. Other elements are similiar to Carbon, but they are not as crucial for life as Carbon.
Link: http://en.wikibooks.org/wiki/AP_Biology/The_Chemical_Building_Blocks_of_Life
LikeLike
It is correct for people to say that we are made out of stardust. Stars owe their light to the energy released by nuclear fusion reactions. These reactions created chemical elements like carbon or iron which are the building blocks that make up the world around us. When a star explodes during a supernovae, the resulting high energy environment enables the creation of some of the heaviest elements. This explosion disperses the different elements across the Universe, and scatters stardust which makes up planets including Earth.
Here is a link that explains how stars come to be and how they connect with some of the most important elements that make up the building blocks of the world around us:
http://www.physics.org/article-questions.asp?id=52
LikeLike
While seeing the video of Carl Sagan explaining an experiment that is 60 years old I looked up the first time this experiment was done in the 1950s by Stanely Miller and Harold Urey. While working on the University of Chicago they conducted this experiment on the origin of life. The experiment they executed was the same one explained in the video and it concluded that as much as 10-15% of carbon was now in the form of organic compounds. Two percent of cabron formed amino acids which are essential to cellular life and used to create proteins. After seeing this video it made me research more about the past experiment and I realized how important carbon really was. Carbon is the back-bone of life on Earth and without it life would simply not exist.
LikeLike
Wow, graphine really is a super compound! The video mentions many things that can be done with graphine in the near future but if you really think about it the possibilities are endless. It makes me wonder how many other unknown compounds and elements are out there and what the future might hold. Who knows, if graphine is such a great discovery I wonder what will come next?!?! I think that its just marvelous how scientists keep discovering new things everyday. Somewhere in the mid 1500’s the world experienced the Scientific Revolution a period of time where new discoveries and theories began to change the world. I believe that right now in the 21st century we are at the beginning of a new era of discoveries and that this is the start of the Second Scientific Revolution. I can’t wait to become a part of it!
LikeLike
Check out this link that the MIT Technology Review published in August:
http://www.technologyreview.com/news/518606/printed-graphene-transistors-promise-a-flexible-electronic-future/
LikeLike
Great link!!! They just keep finding positive things about graphene. Its fun to think about someday having a little flexible computer chip for a cell phone.
LikeLike
Something that really impressed me from this post was the discovery of the planet 55 Cancri e, better known as “the diamond planet”. It is only two times bigger than the Earth but it has eight times its mass. Considering the factors of the temperature it has plus the presence of carbon it makes the ideal conditions for the making of diamonds.
For science fiction it has been a dream to find such interesting astronomic bodies and knowing that one has been discovered, gives great pleasure and what its even more interesting is that it is relatively close to Earth. Also, it has a huge amount of carbon, much more than our sun! It is considered a planet of a whole new class:
“While it’s still unknown exactly what implication this will have on our understanding of evolution of planetary systems, there’s no doubt it is an important step towards understanding the full diversity of planets.”
The discovery of this new planet clearly shows that there is a complete outside world just waiting to be discovered and I know that with all the new technology we will be getting closer and closer to knowing what is really out there.
http://news.nationalgeographic.com/news/2012/10/121011-diamond-planet-space-solar-system-astronomy-science/
LikeLike
The diamond planet orbits a neutron star known as a pulsar. So, here we have a solar system with a single planet—a solar system that can fit inside our sun. And it is a very poetic one:
A diamond the size of a planet, that orbits a dying star.
LikeLike
In this blog post, there is shown a spiral which contains the evolution of how life came to be. As you said, this spiral is ubiquitous, or appearing or found everywhere. Making reference to another blog post of Robert Hooke and how he said and believed that art and science are co-dependent, interdependent,and can be used to describe science or biology in a different perspective. How it contributes to our better understanding of biology and how we can interpret it in many different ways. As you said in the blog post of Robert Hooke and in class, the arts influenced a lot in biology, therefore this also includes literature. I also believe that this spiral is found everywhere. When I first thought about it, I thought about the Nautilus shell. As I researched more about it and how it is relevant to this topic of the spiral and evolution, I found that the chambered nautilus when it is outgrowing its shell it closes one of its “chambers.” This means that the mollusk is ready for a new stage in its life (shell). I also consulted with my mother about the topic of the spiral, and she suggested a poem called “The Chambered Nautilus”, by Oliver Wendell Holmes, that depicts the nautilus shell as continuous, meaning that it outgrows itself and once it leaves its past behind it doesn’t look back but keeps moving forward. I believe this has to do a lot with evolution and the way that it moves forward with things that overcome the last. This also proves how the evolutionary spiral swirls or coils upwards, proving that the emergence of evolution makes changes for the better. Proving that the spiral is ubiquitous it is also seen in our fingerprints, how each and every one of us has a different DNA, therefore being unique and having different sets of fingerprints. This is only possible through evolution.
If you would like too see one of the ways in which the spiral is ubiquitous, I have posted a link of a video of the poem that I mentioned earlier, which shows the chambered nautilus. http://youtu.be/OzC7nMDEiiQ
LikeLike
The spiral is an intriguing natural event that has inspired a lot of art and science.
Take the example of Sanskrit prosody–‘prosody’ refers to rhythm and sound used in poetry (I’m willing to assume that Holmes was somewhat aware of this). The sanskrit oral tradition uses meter–in a symmetric pattern–that the italian mathematician, Leonardo Fibonacci (1170-1250), discovered about one thousand years later, thousands of miles away. This recurrent pattern or sequence is known as the Fibonacci series. And the graphic representation of this sequence is, you guessed it, a spiral known as the golden spiral:
This pattern occurs all throughout the cosmos; including galaxies, hurricanes on Earth, hurricanes on Saturn, the fruit sprouts of a pineapple, the flowering of artichoke, etc. (These latter plant examples are what is known in botany as phyllotaxis).
Great comment and great link, Sofía. I loved the poem. If you have further interest in repetitive, mathematical, patterns in nature, google the concepts ‘fractal’ and ‘broccoli’. You will not be disappointed.
LikeLike
I was not aware that carbon was so important for us to live since by reading this and listening to your class I noticed that almost everything contains carbon. This is why carbon is referred as “the backbone of life”. SPONCH are the six elements that are needed for life to happen because every organism contains them or at least almost all of them to live.
Here explains exactly why carbon is important for us to live and for live to happen.
https://www.boundless.com/biology/organic-chemistry/carbon-and-its-biological-importance/carbon/
LikeLike
When I was first introduced to C.H.N.O.P.S. in 10th grade with Mrs. Rivera, I didn’t understand why phosphorus such a crucial element needed for the existence and maintenance of life on Earth. The scarcity of phosphorus on the Earth’s surface, where much of the phosphorus is locked up in certain minerals that life has difficulty making use of, doesn’t support the fact that it is completely necessary for life on Earth. After revisiting the topic this year I wanted to clear up all the doubts I had about the importance of phosphorus. This link explains the relation this element has to the sustainability of life on Earth. http://www.astrobio.net/exclusive/4968/lifes-first-taste-of-phosphorus-
LikeLike
This quote perfectly resumes the importance of phosphorus for life on Earth:
LikeLike
Until a few weeks ago, I was unaware of how versatile carbon is and that it has an incredibly important role in life. It is present almost everywhere and thanks to its four valence electrons, it can bond with almost any element.It is also the only element that can form strong, stable bonds with itself.
LikeLike
Carbon is an amazing element. And it is the 6th most common substance on the universe.
LikeLike
I found this post very interesting. It really amazes me how we can do so much stuff with a single element. For example graphene, before watching that video I had no idea how much stuff we were able to do with graphene.
I researched more about CHNOPS and the importance of each element to human life and it is very intriguing. It is really amazing to see the different functions of carbon, for example, and the many attributes it has for human life to be possible.
LikeLike
Brief essay on the pattern of spirals in nature:
Nature is full of different shapes and motions seen throughout the passing of time. As scientist progress in new discoveries, one can see how many of these shapes are similar and characterized in different aspects of our nature. The spiral is one of these shapes one gets to see in different activities and materials in Earth and outside.
The principal spiral known in nature is the evolutionary spiral, where one gets to see the development of Earth and its organisms throughout billions of years. This spiral shape can be seen in different quantities and sizes, ranging from the smallest objects, to the biggest of processes. One of the smallest spiral shapes in nature can be found in DNA. This small spiral is produced in large quantities and is responsible for the genetic instructions of each and every human. Ranging into bigger spiral motions, we can see many of nature’s phenomena having this characteristic, like the tornado, forcing large amounts of wind to spin in this twisting motion. This is also seen in a hurricane, where the same forces spin wind and rain in a cycled motion. Even bigger than these spiral processes, we can see many galaxies from outer space having this same shape. Obviously, galaxies are one the biggest spirals in nature, but they also differentiate in sizes.
Throughout time the spiral has proven itself to be one of the most prominent and repeated patters and shapes in our nature, making it an important aspect to the development and progress of our Earth.
LikeLike
Excellent essay, Marie. I encourage you to google “Mandelbrot Set”; another set of shapes and patterns that constantly repeat in nature.
LikeLike
there are 92 elements that are known. 25 of this that are essential for life and six of these elements termed to be the “building blocks of life.” They are, in order of least to most common, sulfur, phosphorous, oxygen, nitrogen, carbon, and hydrogen. The remaining 19 elements are defined as trace elements, which are needed in small quantities, and are usually required by specific proteins to function.
LikeLike
Reading this post helped understand the basic components of life. We are all formed by trillions of small particles that work together as a whole to form living beings. These components were found first in stars and traveled to our Earth in meteors. Throughout millions of years they evolved to form unknown numbers of species that share the same planet. It makes one reflex on the history of the Universe. Life did not start the day we were born, it has been present since centuries before us. But right now we are the most complex form of it on Earth. This should make human beings think about their roles on life and the importance on keeping our evolution and knowledge forward. The link below describes these elements and gives essential information about them.
http://en.wikibooks.org/wiki/AP_Biology/The_Chemical_Building_Blocks_of_Life
LikeLike
“With great power comes great responsibility.” I’m quoting Spider-Man’s uncle, Ben. And I believe he’s correct. Our power is the fact that we are aware our existence. And our responsibility lies in us being the stewards of Earth; which does not mean that we «own» the planet; we are its caretakers.
By learning about our role in the universe, which in many ways means to understand our very essence, we are doing a lot in terms of fulfilling our roles as stewards of this fragile, lonely and blue planet.
LikeLike
It is amazing how even though we may think of ourselves as completely different from our universe, we are composed of the same basic elements. I had never really grasped the importance of the basic elements until I saw this video. It is amazing how interconnected our universe really is. But, if we share these basic elements, why are we the only known planet with intelligent life? I was particularly interested by the Drake equation and researched what it means.
The Drake equation attempts to define the unknown. This equation attempts to estimate the number of technological civilizations in our portion of the cosmos (the Milky way). This equation is written:
N = R* • fp • ne • fl • fi • fc • L
(f sub p; n sub e; f sub l; f sub I; and f sub c)
N = The number of civilizations in The Milky Way Galaxy whose electromagnetic emissions are detectable.
R* =The rate of formation of stars suitable for the development of intelligent life.
fp = The fraction of those stars with planetary systems.
ne = The number of planets, per solar system, with an environment suitable for life.
fl = The fraction of suitable planets on which life actually appears.
fi = The fraction of life bearing planets on which intelligent life emerges.
fc = The fraction of civilizations that develop a technology that releases detectable signs of their existence into space.
L = The length of time such civilizations release detectable signals into space.
This equation is useful for stimulating the intellectual curiosity of our mysterious universe. This equation has no definite answer, no definite variables.
LikeLike
The Drake Equation is basically a thought experiment. Even though there is no definitive answer to it, it is still very useful to ponder the very big–or very small, depending on what values we decide to use i the equation–probability of the human race not being alone in the universe.
LikeLike
Watching the graphene video, I’m still shocked that the product isn’t widely available to the public yet. I’m fascinated by the contradictory nature of graphite, it’s only one atom thin, yet it’s stronger than steel and it would take an elephant standing on a pencil to break it (the elephant thing is something I saw on the internet, not sure if that one is true but it’s an interesting thought.) It is the stiffest known material, yet relatively flexible and can stretch up to 20% of its width. And it’s also the most impermeable material, only allowing water to pass through. I think it’s very interesting that such a seemingly insignificant material is really on if the strongest and most useful on earth. : http://www.zdnet.com/the-10-strangest-facts-about-graphene-3040093050/
LikeLike
That comment was posted at around 8:30pm btw, not 12:22 am
LikeLike
Noted.
LikeLike
It is the strongest material known to man.
LikeLike
I didn’t know that such common elements were so significant in the creation of life in the universe. This post helped me understand why the things we were studying in class were so important. I never would have thought that such a simple element like carbon was the “backbone of life”. But now I understand that carbon is very versatile and it can bond with many elements because of this.
LikeLike
Carbon, of these six elements, is the most versatile. The main reason is that it binds to many elements to form differen compounds. That and it has different forms with individual uses. Diamods, one of it’s form, precipitates in planet Neptune, basically there, it rains diamonds. Why is this important? Well I believe that if technology advances enough get to Neptune, large amounts of diamonds can be retrieved to make structures like indestructible buildings and defense systems being a huge advance against any disaster.
This explains the diamond rains in Neptune.
http://www.spacedaily.com/news/carbon-99d.html
This picture is where I got the idea from.
http://kenobi-wan-obi.tumblr.com/post/6043362
LikeLike
Imagine: “raining diamonds”. We know so little about the Cosmos, that what we may regard as science fiction could actually be science fact.
LikeLike
Biology is a very detailed study of life. It shows us how life is formed, starting from the simplest form of matter and working its way up to the most complex multicellular organisms. All the data that has been gathered during the years is organized according to their role in the composition of life in charts and other models. A spiral can be used as a metaphor to show the emergance of life because in it we see reflected the process that life undertakes as it grows. Molecules combine to build organelles, which then form cells, which continue to join. The result of all these processes is a huge diversity in life (shown by the outermost part of the spiral). Thanks to all the discoveries made by scientists such as biologists, we have a broad understanding of how all life is interconnected.
There is a deeper explanation of how biology and emergence are related in this link: http://www.nature.com/scitable/topicpage/biological-complexity-and-integrative-levels-of-organization-468
LikeLike
I emphasize on emergence because next semester we’ll talk a lot about evolution. And to talk about evolution—or genetics—without a proper grasp on «emergence», is very hard.
Great find, Mariangel.
LikeLike
Thank you!
LikeLike
Pingback: Science Club Post: Starstuff | The Hypertextual Lounge